Aflatoxins are a group of toxic compounds produced as secondary metabolites by fungi Aspergillus flavus and Aspergillus parasiticus. These fungi invade several agricultural commodities and under certain conditions produce these toxins. Although aflatoxins do not affect the crop productivity as such but contaminates the produce, which has deleterious effects on human and animal health. Thus, the aflatoxins, if present in food commodities, reduce their quality and hence the market value.
Among 20 different types of aflatoxins identified, major ones are B1, B2, G1 and G2. Aflatoxin B1 is normally predominant in amount in laboratory cultures as well as in food products. A. flavus typically produces B1 and B2, whereas A. parasiticus produce G1 and G2 as well as B1 and B2 (Aflatoxin G1 and G 2 are produced exclusively by A. parasiticus). Four other aflatoxins M1, M2, B2A, and G2A which may be produced in minor amounts were also subsequently isolated from cultures of A. flavus and A. parasiticus. A number of closely related compounds namely aflatoxin GM1, parasiticol and aflatoxicol are also produced by A. flavus. Aflatoxin M1 (4-hydroxy derivative of aflatoxin B1) and M2 are major metabolites of aflatoxin B1 and B2 respectively, found in milk of animals. These aflatoxins find their way in the milk if the animals are fed on diet contaminated with aflatoxins.
Typical occurrence ratios for aflatoxins B1 and B2 (mainly produced by A. flavus) average approximately 4:1. Typical occurrence ratios for aflatoxin B1 and the sum of the aflatoxins B2, G1 and G2 (the G toxins are mainly produced by A. parasiticus) average approximately 1:0.8, although variations do occur for both ratios.
Aflatoxins are carcinogenic and cause cancer of liver. It lowers the body’s immune response to invasion by foreign substances and acts synergistically with hepatitis viruses B and C. Indian childhood cirrhosis, a liver disorder found among children is caused by aflatoxins. The risk of cancers due to aflatoxin exposure is based on cumulative lifetime dose. Usually large doses or long-term exposure to low levels in the diet have significant adverse effects. Aflatoxin consumption by livestock and poultry causes the disease aflatoxicosis. As liver is the chief site of aflatoxin metabolism, consumption of high amounts of aflatoxins can lead to acute liver disease or even death within 72 hour. Presence of aflatoxins in low concentrations in diet of farm animals results in decreased feed efficiency, loss of weight, reduced egg production and contamination of milk.
Aflatoxins are so toxic that they can exert their harmful effects even if present in microgram quantities in the diet. Considering the extremely toxic nature and the practical problems associated with production and maintenance of food and feedstock completely free from aflatoxins, limits of tolerance have been prescribed for various foodstuffs for human and animal consumption.
Biosynthetic pathway
A Type-I polyketide synthase (PKS) and a pair of specialized fatty acid synthase subunits (FAS-1 and FAS-2) form a complex (1.3 MDa) to synthesize norsolorinic acid. Then a sequence of about 15 redox reactions ensues, often mediated by cytochromes P450. The broader superfamily of these oxygenases is well-known to play central roles in secondary metabolism, and they are seen to figure prominently in the intermediate steps of aflatoxin biosynthesis to carry out inter alia two unusual skeletal cleavage and rearrangement reactions. The biosynthetic pathway progresses as under:
Norsolorinic acid (NOR) →Averantin (AVN) →5'-hydroxyaverantin (HAVN) →Oxoaverantin (OAVN) →Averufanin (AVNN) →Averufin (AVF) →Versiconal hemiacetal acetat (VHA) →Versiconal (VAL) →Versicolorin B (VERB) →Versicolorin A (VERA)→Demethylsterigmatocystin (DMST)→Dihydrodemethylsterigmatocystin (DHDMST) →Sterigmatocystin (ST) →Dihydrosterigmatocystin (DHST)→O-Methylsterigmatocystin (OMST)→Dihydro-O-methylsterigmatocystin (DHOMST)? Aflatoxin B1 (AFB1)→Aflatoxin B2 (AFB2) →Aflatoxin G1 (AFG1) →Aflatoxin G2 (AFG2).

Fig. 1 Schematic diagram of aflatoxin synthesis pathway
All the genes that have been shown to be involved in the biosynthesis of aflatoxins (AF) are located within a 70 kb large region of the A. flavus and A. parasiticus genomes. These identical gene clusters consists of 24 genes of which 20 are involved directly in the biosynthesis of AF by catalyzing one or more steps. The remaining four genes are transcriptional regulators and AF-pumps.
Mechanism of action
The chief site of action is liver cells. Action of AFB1 begins with the ingestion of contaminated food and proceeds through the following four steps:
- Metabolic activation: The cytochrome P450 (CYP450) isoenzyme system in the liver metabolizes aflatoxin B1 (AFB1). The CYP450 catalyze the epoxidation of AFB1 to yield active forms known as AFB1-8, 9-exo-epoxide and AFB1-8,9-endo-epoxide. It is the AFB1-8, 9-exo-epoxide which exerts hepatocarcinogenic effects of AFB1. AFB1 metabolism also results in the formation of aflatoxins Q1, M1, and P1. These metabolites as well as other naturally occurring aflatoxins (G1, B2 and G 2) are poor substrates for epoxidation and, consequently, are less mutagenic, toxigenic, and carcinogenic than AFB1.
- DNA modification: AFB1-8,9-exo-epoxide is highly unstable molecule which has high affinity for guanine (G) bases in the DNA. It binds to the N7 position of guanine through a covalent bond and forms AFB1-N7-guanine adducts in the target cells. These adducts induce DNA mutations primarily through a G to T (thymine) transversion in the DNA.
- Cell deregulation: The G to T transversion (AGG to AGT) at the third base of codon 249 of the protein 53 (p53) tumor suppressor genes in the human liver induces changes in the coding properties of the DNA. The p53 gene is a transcriptional activator which functions as a controller of the cell cycle, and has a role in the apoptosis pathway and DNA repair. The mutations in p53 may cause permanent transcriptional changes as well as interference with RNA synthesis. This result in DNA damage, development of DNA lesions, and consequently cell deregulation.
- Development of hepatocellular carcinoma: Loss of function of p53 give cells a selective growth advantage, since p53 is a tumor suppressor gene. Therefore, the deregulation of p53 leads to tumor formation in the liver known as hepatocelluar carcinoma (HCC).
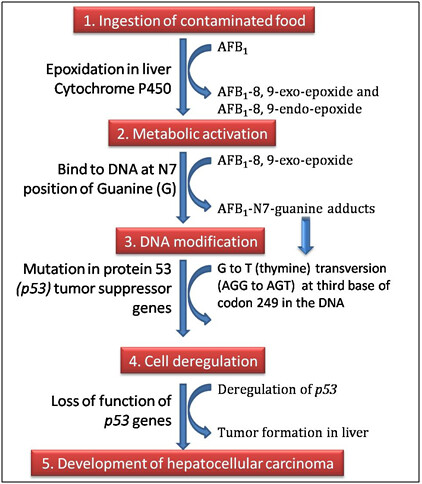
Fig. 2. A simplified diagram of mechanism of action of aflatoxins
References:
Kumar V, Ghewande MP and Basu MS (2005). Safeguard groundnut from aflatoxin contamination. NRCG, Junagadh, Gujarat, India, p. 13.
About Author / Additional Info:
Author is a Senior Scientist (Plant Pathology) working at ICAR-National Research Centre on Litchi, Muzaffarpur, Bihar (India). Currently working on diseases of litchi (Litchi chinensis), and mycorrhizal association in litchi. Formerly, he had worked on groundnut (aflatoxins and disease management) at ICAR- Directorate of Groundnut Research, Junagadh, Gujarat (India).