Biological methods of hydrogen production mainly include: Bio-photolysis of water (Direct or Indirect), Photo-fermentation, Dark-fermentation and Microbial fuel cell. Microbial consortia produce hydrogen by photo/dark fermentation. Most widely studied ways for biological hydrogen production discussed below;
(1) Photo fermentation: Photosynthetic microorganisms (e.g. algae and photosynthetic bacteria) ferment organic substance in presence of light. They either follow oxygenic (Chlamydomonas reinhardtii), or anoxygenic (Rhodobacter sphaeroides) pathway. Hydrogenase and Nitrogenase enzymes are involved in the production of hydrogen by photo (light) fermentation.
(2) Dark fermentation: Facultative anaerobes (Escherichia coli and Enterobacter spp.), or obligate anaerobes (Clostridium spp. including, C.pasteaurianum, C.butyricum, and C.beijerinki)) grow on sugar (as a carbon source) present in waste organic matter and produce hydrogen. Dark fermentative microbes follow pyruvate formate lyase (PFL), and pyruvate ferredoxin oxidoreductase (PFOR) pathway. Theoretically 4.0 mole of hydrogen could be produced in expense of per mole of glucose, called Thauer limit.
Theory of Hydrogen production by Dark-fermentation
Obligate anaerobes are going through PFOR pathway (Acetate or butyrate and hydrogen production). In Acetate and butyrate pathway respectively acetate and butyrate produced as sole by-product. Acetate pathway could produce 4.0 mole of hydrogen/mole of glucose whereas butyrate could produce 2.0 mole of hydrogen/mole of glucose. Reason behind this difference (mole of hydrogen produced/mole of glucose) is that in case of butyrate production, NADH (that produced in glycolysis) is used for the oxidation of aceto-acetyl CoA to butyrate and in the acetate pathway this NADH used for the production of additional hydrogen. Thus, by acetate production (as sole by-product) more hydrogen could be produced in contrast to butyrate (as sole by-product). Acetyl CoA act as major intermediate for both acetate and butyrate pathway.
Facultative Enterobacters produce hydrogen via PFL and FHL (Formate hydrogen lyase). In this route, formate produced as sole by-product that catalyzed by PFL and further degraded into hydrogen and carbon via FHL. PFL also reported in some Clostridia sp.
Hydrogenase
Hydrogenase enzyme plays major role in the reduction (evolution) and oxidation (uptake) of protons that are generated during fermentation of sugars. Foremost study of hydrogenase were carried out on E. coli. There are three types of hydrogenase viz. [Ni-Fe], [Fe-Fe] and [Fe] hydrogenase. [Ni-Fe] and [Fe-Fe] are playing dynamic role in proton uptake and evolution respectively. [Fe-Fe] hydrogenase is extremely sensitive to oxygen but having higher rate of hydrogen evolution (up to 100 fold as compare to [Ni-Fe] hydrogenase). Significant part of [Ni-Fe] hydrogenase has more sensitivity to oxygen and this attribute makes it potential towards metabolic and genetic modifications/engineering.
Two step fermentation
Approach for enhancing biohydrogen production was made by integrating dark and photo fermentation. In first step, anaerobic/facultative anaerobic microbes produce organic acids, hydrogen and carbon dioxide by dark fermentation of lingo-cellulosic biomass. In the second step, under anaerobic condition photosynthetic microbial strains used these organic acids (act as electron donor). Additional hydrogen could be produced by photo-fermentation (in second step) that utilizes dark fermentation effluent-slurry (contains organic acids with high Chemical oxygen demand) to produce further hydrogen.
Overview of two step fermentation is depicted below (Figure 1):
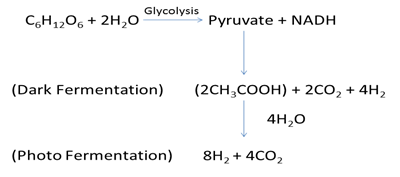
Figure 1: Two step fermentation- When microorganisms metabolize glucose (via glycolysis), NADHs are produced and it is oxidized (NAD+) further for producing organic acids and hydrogen by dark fermentation. These organic acids could further be converted into additional hydrogen via photo-fermentation in second step.
Commercial production of biohydrogen could be established after subduing lower hydrogen yield from cellulosic biomass. In order to overcome this problem, significant effort has been made towards genetic modification of strains, blocking acid and alcohol pathways to enhance NADH production (this NADH further oxidized to produce hydrogen), hydrogenase engineering and others.
References:
http://energy.gov/eere/fuelcells/hydrogen-production-natural-gas-reforming (Accession: 21/11/2015, 07.35pm)
M. Stephen and L. H. (1931). Stickland, Biochem. J., 25, 205.
Ntaikou, I., Antonopoulou, G., Lyberatos, G. (2010). Biohydrogen production from biomass and waste via dark fermentation: A Review. Waste Biomass Valor. 1, 21-39.
Knappe, J., Blaschkowski, H.P., Grobner, P., Schmitt, T. (1974). Pyruvate formate lyase of Escherichia coli: the acetyl enzyme intermediate. Eur. J. Biochem. 50, 253â€"263
Thauer, R.K., Jungermann, K., Decker, K. (1977). Energy conservation in chemotrophic anaerobic bacteria. Bacteriol. Rev. 41, 100â€"180.
Wood, N.P., Jungermann, K.A. (1972). Inactivation of the pyruvate formate lyase reaction of Clostridium butyricum. FEBS Lett. 27, 49â€"52.
Zhu et. al. (2011). In sustainable production of fuels, chemicals, and fibres from forest biomass, ACS symposium series; American Chemical Society, Washington, DC.
About Author / Additional Info:
Research Scholar,
Department of Biotechnology,
National Institute of Technology, Raipur.
Research Interest: Bio-fuels and Biological waste water treatment