It is a process by which haploid or diploid somatic cells develop into structure that resemble zygotic embryo (i.e. bipolar structure without any vascular connection with the parental tissue) through an orderly series of characteristic embryological stages without fusion of gametes. It can also be defined as the process by which the somatic cells or tissues develop into differential embryos and each fully developed embryo is capable of developing into a plantlet.
Somatic embryo formation takes place in two phases i.e induction phase and expression phase. In first phase (induction phase) somatic cells acquire embryonic competence and proliferate as embryonic cells. In second phase (expression phase) cell show and express their embryonic competence and differentiate into embryonic cells. Embryonic competence is the relative ease of inducing embryogenesis and the competent cell is that which has reached transitional state and has already started to become embryonic but still require exogenous stimuli.
Embryonic cells can be explained as the cells that have completed their transition from somatic (non-embryogenic) state to one in which no further exogenously applied stimuli such as application of growth regulators are necessary to produce somatic embryos. Superficially embryogenic cells resemble meristematic cells, they are generally smaller, more isodiametric in shape, have more densely staining nuclei and nucleoli and have denser cytoplasm. Isodiametric is a word that describes parenchyma cells. "Iso" means same and diametric has to do with the dimensions of the cell. Thus, isodiametric literally means "same dimensions." The dimensions of the cell are the length, width, and height. Thus, it means that the length, width and height are approximately equal in each of the parenchyma cells.

Requirements for Initiation of Somatic Embryogenesis
Explants
Any part of plant which is directly used to initiate process of tissue culture is known as explant and the plant from which the part has been taken is called mother plant. In Dicot different plant parts such as; young seedling tissues or embryogenic tissue, inflorescence, young roots, petiole, immature leaf, immature hypocotyle of about 3 weeks can be used for initiation of somatic embryogenesis. In Monocots following explants are used;
• Zygotic embryo----young seeds 10-15 days after pollination
• Young inflorescence----1-2cm in length on sterilized medium
• Young leaf----of unexpanded young seedling 1-2 mm
Other basic requirements
In other basic needs the followings are of most importance;
Auxin supply
Auxins are type of hormones and are essential for proper growth of plant or plant parts. In somatic embryos development process auxin play role in two steps. First the callus is initiated and multiplied in medium with high auxin (2,4-D) supply. Secondly this localized group of meristimatic cells called embryogenic clumps (ECs) is differentiated on proliferation media to develop into mature embryos in low auxin (0.01-0.1mg/L) or no auxin supply at all. Among auxins 2,4-D is commonly used but NAA and IBA use vary from plant to plant. In some cases zeatin has also been reported to use in development of somatic embryos.
Nitrogen Source
Nitrogen supply is very necessary in somatic embryogenesis as it is required for initiation and maturation of somatic embryos. It is used as in reduced form such as ammonium salt. For example in carrot high conc. of nitrogen (in the form of ammonium nitrate) is used in MS medium for development of somatic embryos. Sometimes NH4Cl and NH4NO3 both are used for somatic embryogenesis in number of cultures.
Types/Pathways of Somatic Embryogenesis
There are two pathways of somatic embryo formation which are discussed as followings.
1. Direct somatic embryogenesis: It is the formation of embryo directly from a single cell without an intervening callus stage. Although embryo arises by means of the dedifferentiation of a differentiated cell within the explant.
2. Indirect somatic embryogenesis: It is the process of embryogenesis via intervening callus. The embryogenic callus is composed of young embryos (pre-embryogenic masses or globular embryos) and their further development depends upon the duration of the application of the inductive stimulus. If the period is relatively shorter the process will be direct and if it is long then the process will be indirect.
Expression of Somatic Embryogenesis in Different Plant Groups
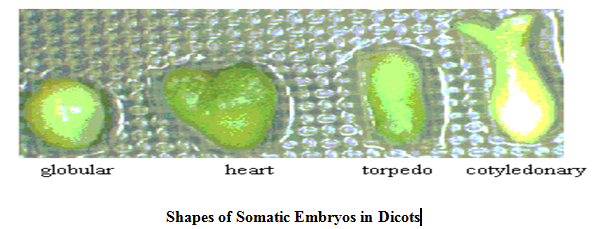
Somatic embryos display their varying expression in different plant groups, like in Monocot these are globular, scutellar (transition) and coleoptilar shape, in Dicot these are pro-embryo, globular, heart shaped, torpedo shaped and bipolar embryo shaped, whereas in conifers these are of globular, early cotyledonary and late cotyledonary shape.
Similarities and Differences between Zygotic Embryos & Somatic Embryos
Both type of embryos originate from single cells, have bipolar structure and do not show vascular differentiation. Somatic embryos differ from zygotic embryos as because of large in size and show more than two cotyledons.
Factors Associated with Somatic Embryogenesis
Explant can be induced to embryogenic cells by variety of procedures/factors that usually include exposure of plant to growth regulators, pH shock, heat shock or treatment with various chemical substances.
Structural Factors:
Structural factors include initiation of polarity, asymmetric division which forms pre-embryos (polarized DNA synthesis), cell size and cell physical isolation.
Physiological Factors:
These are carbon source, NH4, conc., pH change, light intensity & quality, temperature and differences in the contents of reducing sugars & starch (high sugar & low starch).
Gene Expression:
Strategies rely on study of gene and proteins expressed in embryogenic and non embryogenic cells as well as in different stages of embryogenesis. However number of genes specifically expressed is limited & changes of protein pattern is highly regulated post transcriptionally at mRNA level. Hormonal level in culturing medium may modify the synthesis of some somatic embryogenic specific proteins. Various proteins are found during somatic embryogenesis at different levels such as EP1 which is a glycoprotein, present in embryogenic callus and not present in non-embryogenic callus. Similarly EP2 (Lipid transfer protein) present in epidermal cell undergone differentiation during embryogenesis, EP3 (Glycoprotein) which is a temperature sensitive protein that work in proper temperature and at high temperature fails to complete transition from globular to heart shape of somatic embryos. Some other important proteins such as Dc3, Dc8, J4e and ECP31 are also produced at different moments during somatic embryogenesis.
Effect of Exogenously Applied Plant Growth Regulators on Somatic Embryogenesis
Different plant growth regulators applied exogenously in culturing medium showed various expressions in somatic embryogenesis. For example auxin causes differential change in gene expression in pre-embryos and in absence of auxins necessary gene product (mRNA & protein) responsible for globular stage did not produced, ultimately inhibit embryogenic response. Likewise ABA (Abscissic acid) supply also inhibits germination of somatic embryos at later stages.
Effect of Endogenous Hormones (Present in Explants) on Somatic Embryogenesis
Effect of endogenous hormone level in initial explants also varies and it depends upon the concentration of hormones present in explants at initial level. For example if there is more quantity of abscissic acid is present then it will inhibit somatic embryos formation in cultures.
Applications of Somatic Embryogenesis
• Somatic embryogenesis is a very important process of tissue culture and it has lot of applications some important are followings;
• This process can be used for in vitro screening, selection and mass propagation of various plants.
• It is also very helpful to conserve the rare and endangered plant species.
• It can also be applied to obtain pure culture of homogenous material (explants).
• Synthetic seeds are developed by this process which is used for further propagation of those plants in which no natural seed production takes place or flower synchronization/seed formation is not successful.
• This process can be used for protoplast isolation and further studies on protoplast for different purposes.
• A desired gene can be transferred through this process for developing transgenic plants.
About Author / Additional Info:
PhD Scholar