Authors: Vivek K Singh, Kumatkar Raghunath Bhimrao and Pratik I Bhoyar.
CCS Haryana Agricultural University, Hisar, Haryana, 125004.
INTRODUCTION
One of the major concerns of the general public about transgenic crops relates to the mixing of genetic materials between species that cannot hybridize by natural means. To meet this concern, the two transformation concepts cisgenesis and intragenesis were developed as alternatives to transgenesis.
WHAT ARE CISGENESIS/INTRAGENESIS:
Concept of cisgenesis introduced by Dutch researchers Schouten, Krens and Jacobsen (2006). It refers to genetic modification of crops/plants using one of the techniques of recombinant DNA technology, without involvement of any “foreign” DNA; in other words, the manipulation is done using donor DNA fragment from the species itself or from a cross compatible species. The newly introduced gene is unchanged and includes its own introns and regulatory sequences and is free of vector DNA, except T- DNA border sequences that flank the cisgene (natural gene). Therefore, it is not really a new technique. The use of the term is an attempt to distinguish GM plants that contain DNA from unrelated organisms. Intragenesis is very much similar to cisgenesis but the difference lies in the fact that the gene of interest is taken from other source while the regulatory elements and introns from another source and a new combination of DNA fragments are created artificially through in vitro rearrangement. Intragenesis also allows the use of antisense or RNA interference (RNAi) with the aim of silencing the gene (Lusser et al., 2011; Schaart and Visser, 2009).
Classical breeding is crossing of closely or distantly related individuals to produce new crop varieties.
Cisgenesis is genetic modification of a recipient plant with a natural gene from a crossable plant; using its native regulatory elements.
Intragenesis is genetic modification of a recipient plant with genetic elements, such as promoters and coding sequences, from one or more a crossable plants.
Transgenesis is the genetic modification of a recipient plant with one or more genes and/or promoters from any non-plant organisms, or from a donor plant that is sexually incompatible with the recipient plant.
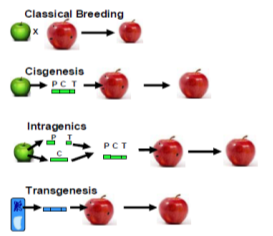
P = Promoter,
C = Coding sequence,
T = Terminator
Table: Major characteristics of different GM concepts
| Characters | Cisgenesis | Intragenesis | Transgenesis |
| Source of gene | Plant itself or from crossable species | plant itself or from crossable species | Could be from any organism |
| Nature of gene | Natural gene | Natural gene | Natural/artificial gene |
| Methods of gene transfer | Mainly Agrobacterium-mediated | Mainly Agrobacterium and some time RNAi | Any methods of recombinant DNA technology |
| Marker gene | Removed | Removed | Contain marker gene |
| Change in gene pool | No change | No change | Change |
WHY CISGENESIS:
Cisgenic plants are probably considered safer than those produced through conventionally breeded plants because of the lack of linkage drag. In cisgenesis, only the desired genes are introduced without the undesirable genes. Cisgenesis furnishes no unnecessary hazard compared to induced translocation or mutation breeding. Therefore, cisgenesis precludes linkage drag, and hence, prevents hazards from unidentified hitch hiking genes (Schouten and Jacobsen 2008). Due to this reason, cisgenesis is normally safe than traditional breeding programmes and various biotic and abiotic stress resistance genes can be pyramided to provide wider and long lasting forms of resistance.
ADVANTAGES OF CISGENESIS OVER CONVENTIONAL BREEDING:
Conquer the setback of linkage drag: In conventional breeding methods, requires a several back crossed generations to overcome the problem of linkage drag. Cisgenesis overcomes the problem of linkage drag and only the gene of interest is introduced into the genome of the recipient plant within a short period of time.
Maintains original genetic make-up of plant variety: In a hybridization method, the genetic makeup of the progeny plant varies from its parents because it has been a mixture of both the parental genomes, whereas in cisgenesis, a new gene has been introduced without changing their genetic makeup.
Reduction in pesticide application: The key purpose of cisgenesis is to transfer disease resistance genes to susceptible varieties. The vital goal here is to lessen substantial pesticide application.
Time saving: Cisgenesis could be employed for the rapid introduction of desired traits into commercially successful cultivars without changing their constructive characteristics.
LIMITATIONS OF CISGENESIS:
Ø Although cisgenics technology is exhibiting considerable advantages over the transgenic counterpart, but still there are a few limitations associated with this technology like; Characters outside the sexually compatible gene pool cannot be introduced.
Ø Sequence information of the plant.
Ø Low transformation efficiencies to create large number of transformants.
Ø The production of marker free plants usually requires the development of innovative protocols, since such protocols may not be readily available for the crop.
SAFETY ASPECTS:
On 16 February 2012, European Food Safety Authority (EFSA, 2012) reported the detail study concerning the safety aspects of cisgenic plants and validated that cisgenic plants are secure to be used in terms of environment, food and feed, similar to the traditionally bred plants. However, the present GMO regulation keeps the cisgenic micro-organisms out from its supervision.
ACHIEVEMENTS:
The first scientific statement of bringing forth a true plant obtained by cisgenic approach was reported in apple through the insertion of the internal scab resistance gene HcrVf2 influenced by their own regulatory genes into the cultivar Gala, a scab susceptible cultivar (Vanblaere et al., 2011). Barley with improved phytase activity was produced successfully by Holme et al. 2011, through cisgenic approach. Late blight resistant potatoes have developed by cisgene stacking of R- gene (jo et al., 2014).
REFERENCES:
EFSA. 2012. Scientific opinion addressing the safety assessment of plants developed through cisgenesis and intragenesis. EFSA J., 10 (2), 1-33.
Holme, I.B.; Dionisio, G.; Brinch-Pedersen, H.; Wendt, T.; Madsen, C.K.; Vincze, E.; and Holm, P.B. 2011. Cisgenic barley with improved phytase activity. Plant Biotechnol. J., 10 (2), 237-247.
Jo, K.R.; Kim, C.J.; Kim, S.J.; Kim, T.Y.; Bergervoet, M.; Jongsma, M.A.; Visser, R.G.F.; Jacobsen, E. and Vossen, J.H. 2014. Development of late blight resistant potatoes by cisgene stacking. BMC Biotech.
Lusser M.; Parisi C.; Plan D. and Rodriguez-Cerezo E. 2011. New plant breeding techniques: State-of-the-art and prospects for commercial development, JRC Scientific and Technical Reports. Luxembourg, European Union, 1-220.
Schaart J.G. and Visser R.G.F. 2009. Novel Plant Breeding Techniques - Consequences of new genetic modificationbased plant breeding techniques in comparison to conventional plant breeding, COGEM Research Report. The Netherlands Commission on Genetic Modification, 1- 60.
Schouten, H.J. and Jacobsen, E. 2008. Cisgenesis and intragenesis, sisters in innovative plant breeding. Trends Plant Sci., 13, 260-261.
Schouten, H.J; Krens, F.A. and Jacobsen, E. 2006. Cisgenic plants are similar to traditionally bred plants: international regulations for genetically modified organisms should be altered to exempt cisgenesis. EMBO, 7, 750-753.
Vanblaere, T.; Szankowski, I.; Schaart, J.; Schouten, H.; Flachowsky, H.; Broggini, G.A.L. and Gessler, C. 2011. The development of a cisgenic apple plant. J. Biotech., 154, 304- 311.
About Author / Additional Info:
I am currently pursuing Ph.D. from CCS HAU, Hisar